Formal experimental record
Aim
To determine what makes ice melt fastest among 4 different household substances.
Hypothesis
The ice cube with salt will melt fastest out of all the dependant variables added to the ice cube.
Materials
• Notebook and pencil
• 5 identical bowls or saucers
• 15 identical ice cubes
• 1 ½ tsp. of Salt
• 1 ½ tsp. of Sugar
• 1 ½ tsp. of Flour
• 1 ½ tsp. of Sand
• ½ teaspoon measuring spoon
• Timer
Method
Step 1. 5 identical ice cubes were placed separately on 5 identical saucers.
Step 2. ½ tsp. of salt, sugar, flour and sand were simultaneously sprinkled over the top of each allocated ice cube.
Step 3. One ice cube (independent variable controlled) was left untouched.
Step 4. Using the timer, the amount of time that each ice cube took to completely melt was timed then recorded.
Step 5. Steps, 1, 2, 3 and 4 were repeated another 2 times.
Results
Conclusion
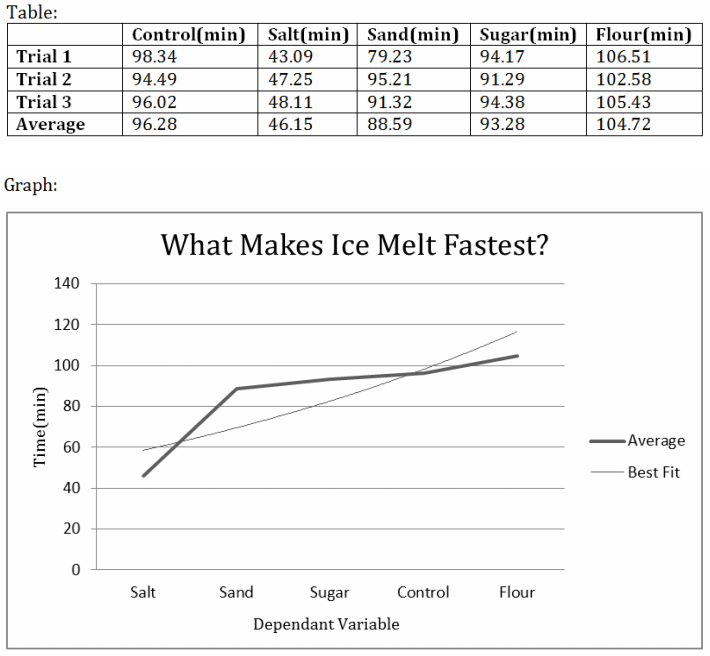
Out of all four household substances added to the ice cube, salt allowed it to melt fastest, at a time of 46 minutes compared to the time of added flour at 104 minutes.
Reflection
One mistake that I made during this experiment was using the ice cubes without knowing if they were completely set or not. I had found out that they were not set when they were melting as there was a clear crater in the centre of the ice cube. Thankfully, there were spare ice cubes in the tray, and I was able to freeze them for longer and continue with the experiment without waiting for another batch of ice cubes to freeze completely. To prevent this from happening next time, I would freeze them for another 2 hours before using it. If I was in a hurry, I would constantly check every 30 minutes for a whitish opaque centre in the ice cube, indicating frozen air bubbles and that the ice cube was completely frozen to the centre.
Also, I could not find a measuring spoon in the house, so I settled with a bottle cap. I searched on the internet and found out that the bottle cap I was using held ½ tsp. of water. So I was able to use an established measuring unit instead of “one bottle cap of”.
If I were to perform this experiment again, I would sprinkle the 4 substances over the ice cube as simultaneously as possible, perhaps getting 5 people, 1 person to place the untouched ice cube down and 4 others to place their allocated ice cubes down and sprinkle the household substances over it. By performing the experiment in this way, I am able to improve and ensure the reliability, validity and accuracy of the experiment.
Also, I would use an actual measuring spoon to conduct this experiment, as it is easier to scoop the substances out of the glass cup and to remove the excess from the top.
Discussion
The knowledge of salt melting ice faster can be applied in a number of real life situations.
One real life situation would be spreading salt on ice in order to melt the snow and ice that has built up on roads in winter. If one has ever seen salt melting ice on the roads, they would immediately see the dissolving process happen. The snow or ice that is covered with the grains of salt immediately melts into water, and the melted ice spreads out from that point. This can be compared to this “What makes ice melt fastest?” experiment, as the exact same process occurred as seen in the pictures above.
Bibliography
• Answers.com, (2014). Can flour melt ice faster than salt?.
[online] Available at: http://wiki.answers.com/Q/Can_flour_melt_ice_faster_than_salt
[Accessed 02 Aug. 2014].
• Answers.com, (2014). How does sand melt ice?.
[online] Available at: http://wiki.answers.com/Q/How_does_sand_melt_ice
[Accessed 02 Aug. 2014].
• Bored of Studies, (2011). Definition of validity, reliability, and accuracy for science. [online] Available at:
http://community.boredofstudies.org/953/general-discussion-2012-hsc/271590/definition-validity-reliability-accuracy-science.html
[Accessed 02 Aug. 2014].
• Day, N. (2014). How Can Sand Melt Ice? | eHow.
[online] eHow. Available at: http://www.ehow.com/how-does_4886477_how-can-sand-melt-ice.html
[Accessed 02 Aug. 2014]
• Helmenstine, T. (2014). Control vs. Experimental Group: How Do They Differ?.
[online] About.com Chemistry. Available at:http://chemistry.about.com/od/chemistryterminology/a/What-Is-The-Difference-Between-Control-Group-And-Experimental-Group.htm [Accessed 09 Aug. 2014].
• Helmenstine, T. (2014). Understand the Difference Between Independent and Dependent Variables.
[online] About.com Chemistry. Available at: http://chemistry.about.com/od/chemistryterminology/a/What-Is-The-Difference-Between-Independent-And-Dependent-Variables.htm [Accessed 10 Aug. 2014].
• HowStuffWorks, (2014). HowStuffWorks "Why do they use salt to melt ice on the road in the winter?".
[online] Available at:
http://science.howstuffworks.com/nature/climate-weather/atmospheric/road-salt.htm
[Accessed 7 Aug. 2014].
To determine what makes ice melt fastest among 4 different household substances.
Hypothesis
The ice cube with salt will melt fastest out of all the dependant variables added to the ice cube.
Materials
• Notebook and pencil
• 5 identical bowls or saucers
• 15 identical ice cubes
• 1 ½ tsp. of Salt
• 1 ½ tsp. of Sugar
• 1 ½ tsp. of Flour
• 1 ½ tsp. of Sand
• ½ teaspoon measuring spoon
• Timer
Method
Step 1. 5 identical ice cubes were placed separately on 5 identical saucers.
Step 2. ½ tsp. of salt, sugar, flour and sand were simultaneously sprinkled over the top of each allocated ice cube.
Step 3. One ice cube (independent variable controlled) was left untouched.
Step 4. Using the timer, the amount of time that each ice cube took to completely melt was timed then recorded.
Step 5. Steps, 1, 2, 3 and 4 were repeated another 2 times.
Results
Conclusion
Out of all four household substances added to the ice cube, salt allowed it to melt fastest, at a time of 46 minutes compared to the time of added flour at 104 minutes.
Reflection
One mistake that I made during this experiment was using the ice cubes without knowing if they were completely set or not. I had found out that they were not set when they were melting as there was a clear crater in the centre of the ice cube. Thankfully, there were spare ice cubes in the tray, and I was able to freeze them for longer and continue with the experiment without waiting for another batch of ice cubes to freeze completely. To prevent this from happening next time, I would freeze them for another 2 hours before using it. If I was in a hurry, I would constantly check every 30 minutes for a whitish opaque centre in the ice cube, indicating frozen air bubbles and that the ice cube was completely frozen to the centre.
Also, I could not find a measuring spoon in the house, so I settled with a bottle cap. I searched on the internet and found out that the bottle cap I was using held ½ tsp. of water. So I was able to use an established measuring unit instead of “one bottle cap of”.
If I were to perform this experiment again, I would sprinkle the 4 substances over the ice cube as simultaneously as possible, perhaps getting 5 people, 1 person to place the untouched ice cube down and 4 others to place their allocated ice cubes down and sprinkle the household substances over it. By performing the experiment in this way, I am able to improve and ensure the reliability, validity and accuracy of the experiment.
Also, I would use an actual measuring spoon to conduct this experiment, as it is easier to scoop the substances out of the glass cup and to remove the excess from the top.
Discussion
The knowledge of salt melting ice faster can be applied in a number of real life situations.
One real life situation would be spreading salt on ice in order to melt the snow and ice that has built up on roads in winter. If one has ever seen salt melting ice on the roads, they would immediately see the dissolving process happen. The snow or ice that is covered with the grains of salt immediately melts into water, and the melted ice spreads out from that point. This can be compared to this “What makes ice melt fastest?” experiment, as the exact same process occurred as seen in the pictures above.
Bibliography
• Answers.com, (2014). Can flour melt ice faster than salt?.
[online] Available at: http://wiki.answers.com/Q/Can_flour_melt_ice_faster_than_salt
[Accessed 02 Aug. 2014].
• Answers.com, (2014). How does sand melt ice?.
[online] Available at: http://wiki.answers.com/Q/How_does_sand_melt_ice
[Accessed 02 Aug. 2014].
• Bored of Studies, (2011). Definition of validity, reliability, and accuracy for science. [online] Available at:
http://community.boredofstudies.org/953/general-discussion-2012-hsc/271590/definition-validity-reliability-accuracy-science.html
[Accessed 02 Aug. 2014].
• Day, N. (2014). How Can Sand Melt Ice? | eHow.
[online] eHow. Available at: http://www.ehow.com/how-does_4886477_how-can-sand-melt-ice.html
[Accessed 02 Aug. 2014]
• Helmenstine, T. (2014). Control vs. Experimental Group: How Do They Differ?.
[online] About.com Chemistry. Available at:http://chemistry.about.com/od/chemistryterminology/a/What-Is-The-Difference-Between-Control-Group-And-Experimental-Group.htm [Accessed 09 Aug. 2014].
• Helmenstine, T. (2014). Understand the Difference Between Independent and Dependent Variables.
[online] About.com Chemistry. Available at: http://chemistry.about.com/od/chemistryterminology/a/What-Is-The-Difference-Between-Independent-And-Dependent-Variables.htm [Accessed 10 Aug. 2014].
• HowStuffWorks, (2014). HowStuffWorks "Why do they use salt to melt ice on the road in the winter?".
[online] Available at:
http://science.howstuffworks.com/nature/climate-weather/atmospheric/road-salt.htm
[Accessed 7 Aug. 2014].
